ArthroFlex®

ArthroFLEX®
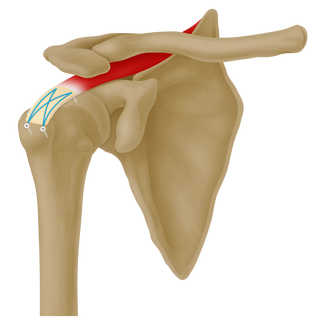
ArthroFlex® is an acellular dermal matrix used to supplement soft tissue repairs such as the Achilles tendon and rotator cuff. It has ≥ 97% DNA removal, comes ready-to-use right out of the package, can be stored at room temperature (15°C - 30°C), and has a Sterility Assurance Level (SAL) of 10-6, which is equal to medical device-grade sterility.1,2
- Improved Clinical Outcomes: ArthroFlex demonstrated decreased re-tear rates and improved patient reported outcome scores after one and two years3,4
- Increased Safety: Sterile with a SAL of 10-6 means lower risk of disease transmission
- Decellularized: Uses Matracell® technology to remove ≥ 97% DNA and cellular remnants to decrease likelihood of immune response1
- Promotes Rapid Healing: Biocompatible and retains natural growth factors, collagen and elastin1
- Convenient: Preservon® technology allows graft to be stored fully hydrated at room temperature (15°C - 30°C) so it can be used right out of the package, without lengthy OR reconstitution processes1,2
- Versatility: Available in multiple sizes and thicknesses
Clinical Application
- Augment repairs of the rotator cuff
- Augment repairs of the Achilles, biceps, quad and patellar tendons
- Reinforce peroneal/tibialis tendons and flexor/extensor tendons of hand and foot
- Reinforce capsular repairs
- Suture reinforcement of soft tissue repairs
| Description | Sizing | |
|---|---|---|
| AFLEX500 | Thickness: 0.5 mm (0.3 - 1.0) | 30 x 40 mm |
| AFLEX400 | Thickness: 1.0 mm (1.26 - 1.74) | 40 x 40 mm |
| AFLEX401 | Thickness: 1.0 mm (1.26 - 1.74) | 40 x 70 mm |
| AFLEX402 | Thickness: 1.0 mm (0.76-1.24) | 20 x 25 mm |
| AFLEX403 | Thickness: 1.0 mm (0.76-1.24) | 25 x 30 mm |
| AFLEX100 | Thickness: 1.5 mm (1.26 - 1.74) | 35 x 35 mm |
| AFLEX101 | Thickness: 1.5 mm (1.26 - 1.74) | 40 x 70 mm |
| AFLEX103 | Thickness: 1.5 mm (1.26 - 1.74) | 50 x 90 mm |
| AFLEX150 | Thickness: 1.5 mm (1.26 - 1.74) | 15 x 140 mm |
| AFLEX200 | Thickness: 2.0 mm (1.26 - 1.74) | 35 x 35 mm |
| AFLEX201 | Thickness: 2.0 mm (1.26 - 1.74) | 40 x 70 mm |
| AFLEX202 | Thickness: 2.0 mm (1.76 - 2.25) | 25 x 30 mm |
| AFLEX822 | Thickness: 2.0 mm | BioWasher® 10 x 14 mm, 2 pack |
| AFLEX300 | Thickness: 3.0 mm (2.5 - 3.5) | 40 x 50 mm |
| AFLEX301 | Thickness: 3.0 mm (2.5 - 3.5) | 40 x 70 mm |
| AFLEX302 | Thickness: 3.0 mm (2.5 - 3.5) | 50 x 60 mm |
| AFLEX352 | Thickness: 3.0 mm (2.50-3.50) | 20 x 30 mm |
| AFLEX600 | Thickness: 4.0 mm (3.7-4.6) | 25 x 35 mm |
| AFLEX601 | Thickness: 4.0 mm (3.7-4.6) | 40 x 70 mm |
| AFLEX602 | Thickness: 4.0 mm (3.7-4.6) | 20 x 30 mm |
| AFLEX650 | Thickness: 5.0 mm (4.7-5.7) | 25 x 35 mm |
| AFLEX651 | Thickness: 5.0 mm (4.7-5.7) | 40 x 70 mm |
| AFLEX652 | Thickness: 5.0 mm (4.7-5.7) | 20 x 30 mm |
References
1.Moore MA, Samsell B, Wallis G, Triplett, S, Chen S, Linthurst Jones A, Quiz X. “Decellularization of Human Dermis Using Non-Denaturing Anionic Detergent and Endonuclease: A Review.” Journal of Cell and Tissue Banking.
2.LifeNet Health. Decellularized Dermis Instructions for Use. 63-0050-01
3.Gilot GJ, Alvarez AM, Barcksdale L. Outcome of large to massive rotator cuff repairs with and without extracellular matrix augmentation: A prospective comparative study. Arthroscopy. 2015 Apr17.pii: S0749-8063(15)00141-3
4.Denard PJ, Brady PC, Adams CR, Tokish JM, & Burkhart SS. Preliminary Results of Arthroscopic Superior Capsule Reconstruction with Dermal Allograft. Arthroscopy. 2018 Jan:34(1):93-99
