ArthroFlex® SCR

ArthroFLEX
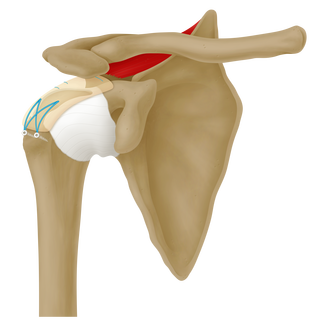
ArthroFlex® SCR is a specific size of acellular dermal matrix designed specifically for the Superior Capsular Reconstruction surgical technique. Biomechanical testing shows the graft restores glenohumeral joint stability and subacromial contact characteristics.1, 2
- Shown to improve patient-reported outcomes: Patients reported decreased pain and improved function scores.7
- Strong: High ultimate load to failure and sutures will not easily pull through graft.3
- Safe: Sterile with a SAL of 10-6.
- Decellularized: Uses Matracell® technology to remove ≥97% DNA and cellular remnants to decrease likelihood of an immune response.4
- Promotes rapid healing: Biocompatible and retains natural growth factors, collagen and elastin.4,5 Addresses most common mode of failure by reinforcing suture-tendon interface.7
- No re-hydration needed: Supplied pre-hydrated and ready for use.5
- Convenient storage: Room temperature (15 - 30˚C).
- Superior Capsular Reconstruction
| Order Code | Description | Sizing |
|---|---|---|
| AFLEX300 | Thickness: 3.0 mm (2.5 - 3.5) | 40 x 50 mm |
| AFLEX301 | Thickness: 3.0 mm (2.5 - 3.5) | 40 x 70 mm |
| AFLEX302 | Thickness: 3.0 mm (2.5 - 3.5) | 50 x 60 mm |
1. Hirahara AM & Adams CR. Arthroscopic Superior Capsule Reconstruction for Treatment of Massive Irreparable
Rotator Cuff Tears. Arthroscopy Techniques. 2015 Dec:4(6):e637-41.
2. Mihata T, McGarry MH, Pirolo JM, Kinoshita M, Lee TQ. Superior Capsule Reconstruction to Restore Superior
Stability in Irreparable Rotator Cuff Tears: A Biomechanical Cadaveric Study. AJSM 2012;40:2248-55.
3. Data on File at Arthrex, Inc. TR-2946.
4. Moore MA, Samsell B, Wallis G, Triplett, S, Chen S, Linthurst Jones A, Quiz X. “Decellularization of Human Dermis
Using Non-Denaturing Anionic Detergent and Endonuclease: A Review.” Journal of Cell and Tissue Banking.
5. Data on file at Lifenet Health. 68-20-048, IFU 63-0050-01.
6. Barber FA, Herbert MA, Boothby MH. Ultimate tensile failure loads of a human dermal allograft rotator cuff
augmentation. Arthroscopy 2008 Jan;24(1):20-24.
7. Burkhart SS. Pranchknum JJ, Hartzler RU. Superior Capsular Reconstruction for the Operatively Irreparable Rotator
Cuff Tear: Clinical Outcomes Are Maintained 2 Years After Surgery. Arthroscopy. 2019.
